Why Are Viral Capsids Icosahedral?

From the late 1980s through the early 2000s, a famous debate played out between the evolutionary biologists Stephen Jay Gould and Simon Conway Morris on the nature of the evolutionary process.
Gould viewed evolution as radically contingent: if “the tape of life” were to be rerun, he argued, even small environmental changes would result in widely divergent outcomes. The likelihood that any major biological innovation, such as multicellularity, photosynthesis, or intelligent life, could evolve again would be vanishingly small, since each of these innovations depended on a combination of rare events.1 The crux of Gould’s idea was that “no important and sufficiently specific evolutionary outcomes are robustly replicable.”
Conway Morris agreed that historical contingencies are pervasive in evolution; however, in his view:
…contingency is inevitable, but unremarkable. It need not provoke discussion, because it matters not. There are not an unlimited number of ways of doing something. For all its exuberance, the forms of life are restricted and channeled.2
In other words, if we imagine life forms having emerged as solutions to problems in a global search space, there are only a limited number of good solutions given existing environmental constraints. Biology is beholden to the laws of physics and chemistry, which limit which solutions are feasible, let alone optimal.
This is why, as species evolve under similar selective pressures toward greater fitness, they acquire similar characteristics. Indeed, even though evolution is contingent at a local level (such as a specific protein sequence or the shape of a flower), it is remarkably predictable at a global level (such as the very existence of proteins and flowers across many species).
Convergent evolution reflects, then, a widespread predictability of life’s design solutions. Similar biological forms and functions often emerge independently in unrelated lineages. For example, ice growth-inhibiting antifreeze proteins evolved independently in Arctic and Antarctic fishes. Although arising at opposite ends of the globe, these proteins converged at both the sequence and structural level: at both locations, they contain a repeating tripeptide motif of glycine–alanine (or proline)–threonine, with the last amino acid fused to a sugar. The severe constraint of survival in freezing seawater pushed these phylogenetically distant fishes to evolve similar molecular solutions.
Similarly, C4 photosynthesis has been documented in more than 60 different plant lineages, including maize, sugarcane, and papyrus. This convergent evolution involves not just a single protein, but a complex mixture of biochemical and structural adaptations that together enable a more efficient use of carbon dioxide.
Nowhere does the power of convergent evolution appear so insistently and compellingly, though, as in the structure of viral capsids, the self-assembling protein shells enclosing viral genomes. Though ranging in size from 20 nm to 800 nm, an estimated 70 percent of viral capsids known to date are icosahedral, shaped like tiny soccer balls.3
This convergence of viral capsid forms is not merely a curiosity of nature. By deciphering viral capsid architecture at the molecular level, scientists have found inspiration for designing more efficient drug delivery vehicles and vaccines. Viral capsid-like protein nanocontainers have provided an excellent solution to the design problem of carrying and delivering a variety of biological cargoes.
But why did viruses converge on icosahedral shapes in the first place? To answer this, we must turn to both genetics and structural biology.
{{signup}}
Evolutionary Constraints
From a genetic perspective, viruses must be economical with how much space in the genome they allocate to encoding capsid proteins, as well as all the other functional genes required for replication and virulence. James Watson and Francis Crick termed this the principle of genetic economy. Such a constraint favors highly symmetrical capsid morphologies so that the same capsid proteins, or a very small number of them, can be used repeatedly for building the capsid, rather than coding for a large number of unique proteins.
For example, the hepatitis B virus has only four genes in its genome,4 of which one encodes the capsid protein. An icosahedron has 60 different rotational symmetrical operations (more than any other platonic solid), and so a capsid with icosahedral symmetry significantly reduces genetic costs.
Second, from a geometric perspective, an icosahedron comes closest to a sphere than other platonic solids.5 This grants it the largest volume for a given surface area or, equivalently, for a fixed number of capsid coat proteins. With such high geometric efficiency, more genomic material can be packaged into an icosahedral capsid than into any other shape, be it a cube, tetrahedron, or octahedron.6
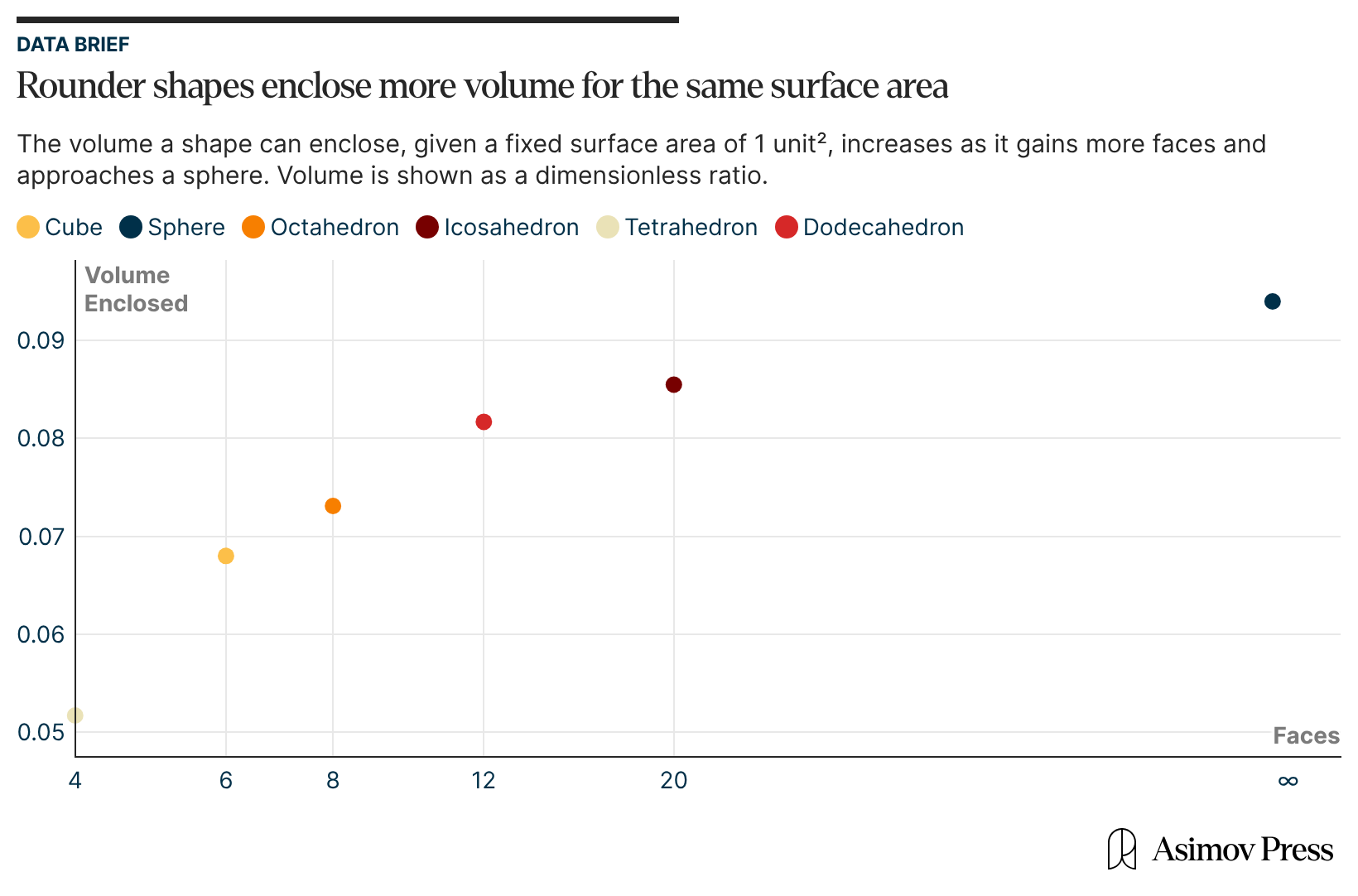
The volume of a viral capsid is important not only because of the size of the viral genome, but also because nucleic acids carry a self-repulsing negative charge that must be physically contained. These repeating negative charges are distributed along the length of the nucleic acid molecules, and in solution, they are surrounded by positively charged ions like Na+and Mg2+ or by more complex molecules like polyamines, which screen the negative charge of the phosphate backbone.
Capsid proteins often display positive charges on their inward-facing surface that bind the genome, thus partially displacing those ions and facilitating capsid assembly and the condensing of the genome inside the protein shell. In viruses with stiff, double-stranded DNA genomes, the bending of the nucleic acid chain imposes an additional internal pressure amounting to many atmospheres.7
Icosahedral capsids are well suited to accommodate these physical constraints because, due to their quasi-spherical shape, they distribute stress more evenly than polyhedra with fewer faces. This becomes especially important when the virus must survive extreme conditions like drying, pH shifts, or mechanical stress.
In addition to its genetic economy and geometric optimality, the icosahedral packaging of viral capsids is also energetically favorable, such that the assembly of many capsids is highly efficient and spontaneous (it even happens in vitro!).8
The Caspar-Klug theory explores this energetic aspect of viral capsid structure. Its inspiration comes from modern architecture, specifically Buckminster Fuller’s geodesic domes. The same principles of structural stability that work for these domes also apply across orders of magnitude of sizes, from molecular to macroscale, and even down to the atomic level.
The story of how this theory came about thus involves not only structural biologists but also architects, artists and chemists.9
The Structural Biology of Viruses
Donald Caspar grew up in Ithaca, New York, in a family of chemists. He first learned about the tobacco mosaic virus (TMV) at the tender age of ten from the renowned crystallographer Isidor Fankuchen, who frequented his family home.
TMV was the first virus ever discovered, in the late 19th century, and its molecular structure was still unknown when Caspar heard about it in the 1930s. However, it was known to plague the leaves of valuable crops, such as tobacco, tomatoes, peppers, causing leaves to curl and the plants, eventually, to die. Understanding its structure, besides being an exciting problem in basic biology, also promised a path toward fighting the virus.
Caspar’s fateful encounter with Dr. Fankuchen decided his professional path — he went on to study TMV and wrote his doctoral dissertation on “The Radial Structure of Tobacco Mosaic Virus,” explaining how this rod-shaped virus had a helical arrangement of capsid proteins along the length of its RNA genome. After completing his doctorate, he moved to the UK in the 1950s to continue his study of viral structures.
Caspar was not the only scientist attempting to solve the structure of TMV. Indeed, many of the greatest thinkers of the time converged at the University of Cambridge because it had the most powerful X-ray sources for crystallographic imaging, crucial for the structural studies of viruses. James Watson was primarily interested in TMV in hopes of solving the structure of RNA.10
Meanwhile, at the University of London, Rosalind Franklin made excellent X-ray diffraction photographs of TMV capsids, albeit without RNA. A fortuitous encounter with these photographs converted another physicist and crystallographer, Aaron Klug, who had just begun his postdoctoral studies in London on a ribonuclease structure but abandoned it in favor of viruses.11
Caspar met Franklin and Klug in 1955, and initially both Caspar and Klug worked independently on different small “spherical” plant viruses, tomato bushy stunt virus (BSV) and turnip yellow mosaic virus (TYMV), respectively. Caspar’s initial analysis revealed a surprising result: the diffraction patterns of the BSV capsid crystals had a non-crystallographic, five-fold symmetry. This pointed to an icosahedral structure, since each of the 12 vertices of an icosahedron is a convergence point of five edges.12
Crick and Watson had previously hypothesized icosahedral symmetry in spherical viruses, but Caspar’s results provided the first experimental evidence for it.13 A little later, Klug and Franklin reported similar results with TYMV. Even though Caspar and Klug worked on similar research projects, their collaboration wouldn’t start until 1958, after Rosalind Franklin’s untimely death.14
The original model of the viral capsid structure suggested by Watson and Crick, in 1956, viewed the capsid as a curved, two-dimensional “surface crystal” (as opposed to the space-filling 3D crystal), closed in on itself and made of identical subunits — capsid proteins. As each subunit was identical, they interacted in the same way, as in true crystals. But such a model only permitted a maximum of exactly 60 subunits, since the icosahedron has 20 triangular faces, and each triangle can be made of three protein molecules.
Advances in electron microscopy of viral capsids, however, particularly the negative staining method,15 revealed that many, if not most, viruses have a more complex structure than could be explained by the Watson-Crick model. They appeared to possess far more than 60 identical subunits per capsid. A new mathematical theory was needed to account for this seeming anomaly. The solution came not from biology, but rather from architecture.

Birth of the Caspar-Klug Theory
John McHale, a London-based avant-garde artist, learned about the structure of the poliovirus (another icosahedral virus Klug studied with his colleague, John Finch) from a newspaper, The Observer. As a member of an art movement that looked for inspiration in science and popular culture (“Independent Group,” a precursor to “pop art”), he had a keen interest in the structures of Buckminster Fuller — by then a renowned, if eccentric, architect.
McHale immediately saw a connection between the icosahedral poliovirus and Fuller’s geodesic domes, hemispherical polyhedra made of rigid triangular elements, which also had icosahedral symmetry. McHale arranged a meeting between Fuller and Klug in July 1959. Despite Fuller’s impenetrable prose, Klug carefully studied his Synergetic Geometry, an unpublished manuscript of what turned into Fuller’s magnum opus. Even with such preparation, their first meeting didn’t result in insights beyond their shared focus on icosahedral symmetry.
In 1960, Caspar and Klug, now separated by the Atlantic, both read a new book by the author Robert W. Marks on Fuller’s work, The Dymaxion World of Buckminster Fuller, and closely studied the structure of geodesic domes. Caspar also met Fuller in person while the latter was serving as a Professor of Poetry at Harvard University in 1962.16
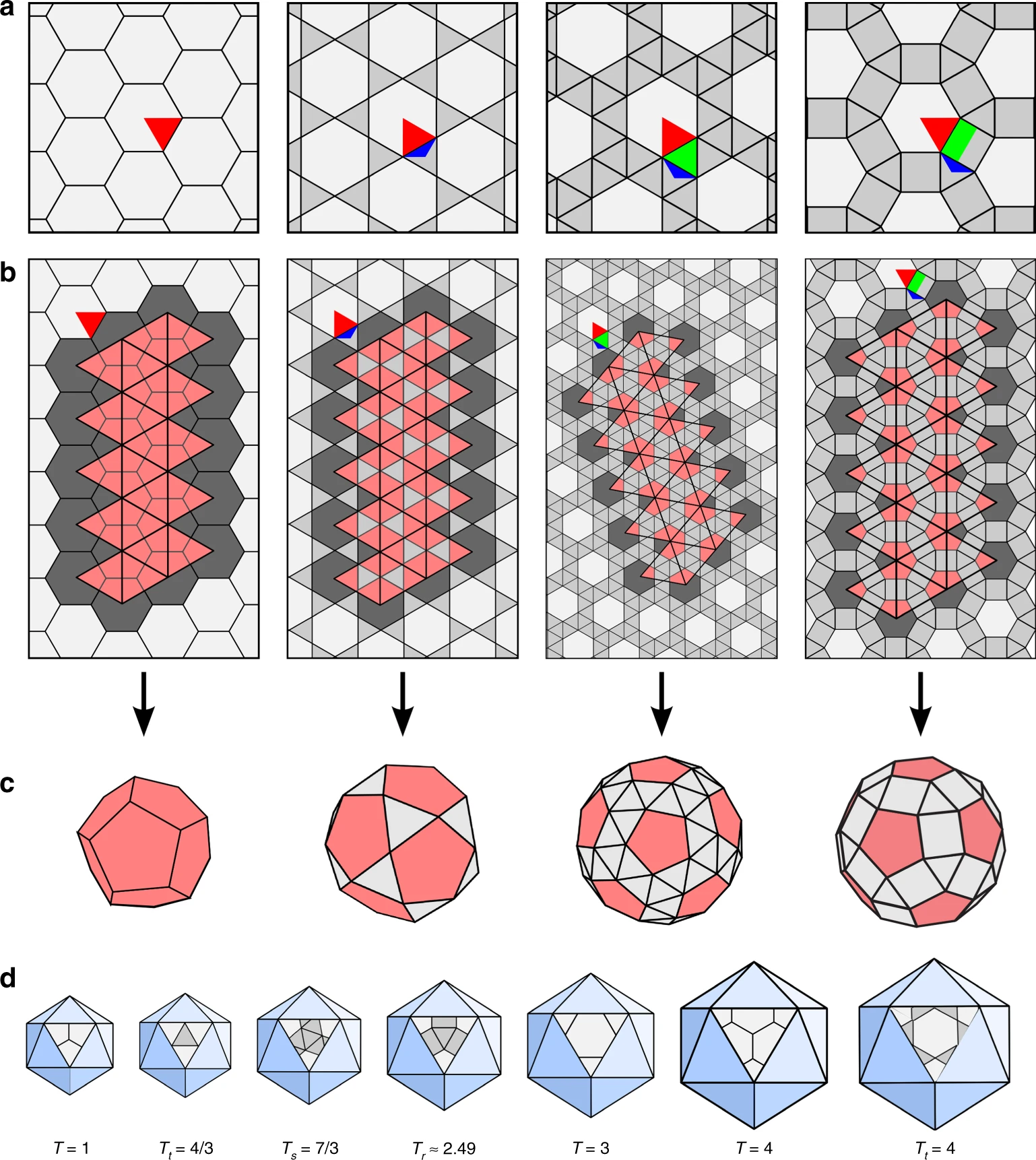
Both Klug and Caspar were particularly interested in the domes’ light yet sturdy structures, consisting of a variable number of hexagons and the mathematically required and invariable 12 pentagons.17 This exact number is required by Euler’s rule to form a closed polyhedron. With only hexagons, the structure won’t close in on itself and will remain flat.
The framework of geodesic domes is made of rigid rods, or struts. Since each of them can be a part of either a hexagon or a pentagon, their connections are not identical due to the different angles between the sides of those two types of polygons. Even so, the interactions between the struts are similar enough across the entire framework to consider them “quasi-equivalent.”


This key concept, introduced by Caspar and Klug, lies at the heart of their theory of viral capsid structures. Like struts in geodesic domes, identical viral capsid subunits could occupy quasi-equivalent local positions, being a part of a hexamer (a ring of six subunits) or of one of the obligatory 12 pentamers (a ring of five). Since the connections between the subunits do not need to be identical, more than 60 subunits per capsid can easily be accommodated, resolving the mystery which Watson and Crick’s model couldn’t explain.
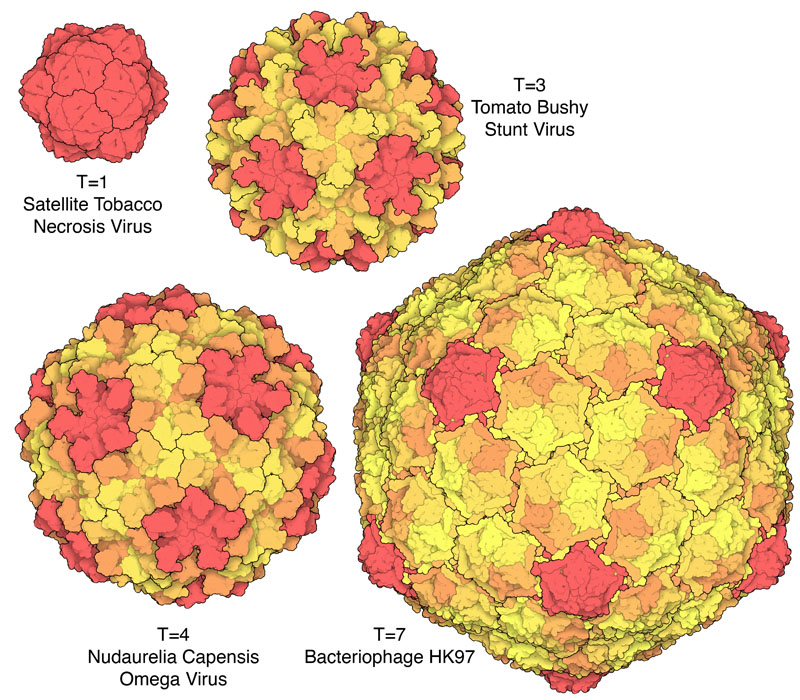
But the Caspar-Klug theory imposed its own constraints on the permissible number of protein subunits in a viral capsid. It introduced a parameter, T, called the triangulation number18: if each face of the icosahedron is divided into triangles (triangulated), then T describes their quantity.19 Since each of these triangles, in turn, can be made up of three capsid proteins, and an icosahedron has 20 faces, the total viral capsid protein numbers thus become “quantized” in multiples of 60 (60T).
The Caspar-Klug theory thus makes a strong prediction: since each triangular face of the icosahedron can accommodate only a certain number of triangles (1, 3, 4, 7…), only certain multiples of 60 are likely to be observed in viral capsids: 60, 180, 240, 420, and so on.20
The pair presented their research in June 1962, at the Cold Spring Harbor Symposium on Basic Mechanisms in Animal Virus Biology, in a now-famous paper titled “Physical Principles in the Construction of Regular Viruses.” In it, they acknowledged that their model of viral capsid structure “was, in fact, inspired by the geometrical principles applied by Buckminster Fuller in the construction of geodesic domes.”21 22 They even included a photo of Fuller’s dome in Montreal, the Biosphère. Besides introducing the principle of quasi-equivalence and the triangulation number, they also coined the term “self-assembly” to describe the spontaneous nature of viral capsid assembly:
Self assembly is a process akin to crystallization and is governed by the laws of statistical mechanics. The protein subunits and the nucleic acid chain spontaneously come together to form a simple virus particle because this is their lowest energy state.23
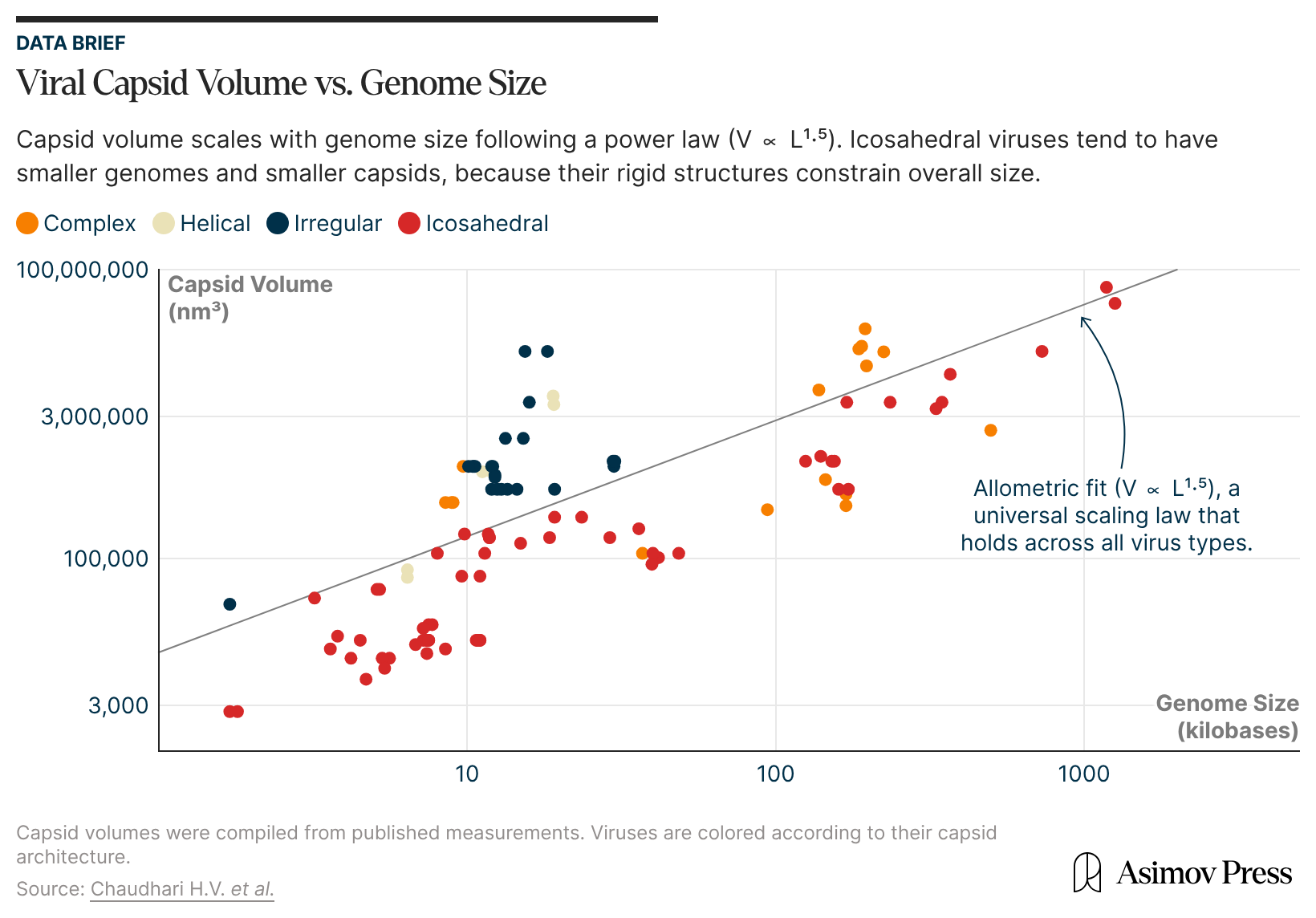
For several decades, the Caspar-Klug theory faithfully described all the known viral structures. But with the development of higher-resolution imaging techniques like cryo-electron microscopy, exceptions soon appeared to the strict constraints on the virus capsid composition (60T subunits) predicted by the theory. The first detected outliers were the cancer-causing polyoma and papilloma viruses, which have capsids made up of 72 pentamer facets.

It wasn’t until the early 2000s that mathematical biologist Dr. Reidun Twarock, at the University of York in England, started working out an expanded mathematical framework to explain these anomalies, called viral tiling theory. It retains the Caspar-Klug theory as a special case but also accommodates a broader range of viral capsid architectures.
Unlike hexagons, pentagonal faces cannot be evenly divided into equilateral triangles; neither can they tessellate, or tile, a 2D plane without gaps or overlaps. To account for this, Twarock turned to Penrose tilings, which can, in fact, tile a plane with five-fold symmetry but with a combination of several distinct shapes; one instead uses “kites” and “darts,”24 or kites and rhombs, all of which are four-sided. Such tilings are non-periodic (made of non-identical types of shapes) but still produce long-range order, as in quasi-crystals. Kites and rhombs represent different types of biological interactions — within trimers and dimers, respectively.
A beautiful mathematical result obtained by Dr. Twarock and colleagues says that these non-periodic tilings, representing flattened capsid structures, can be mapped onto a particular subset of the so-called Archimedean lattices. These are tilings of the plane with a combination of polygons making up a repeating unit (like kites and darts). One of them is the regular hexagonal lattice, and it corresponds to the classic Caspar-Klug construction.
But there are three more tilings within Archimedean lattices that were previously undescribed in the context of viral capsid structures. They can be represented as tilings where hexagons are separated from each other by different sets of repeating polygons of other shapes — triangles, squares, or a combination of the two. Such tilings serve as much better models of more complex viral capsid structures, previously impossible to explain within the framework of the Caspar-Klug theory.
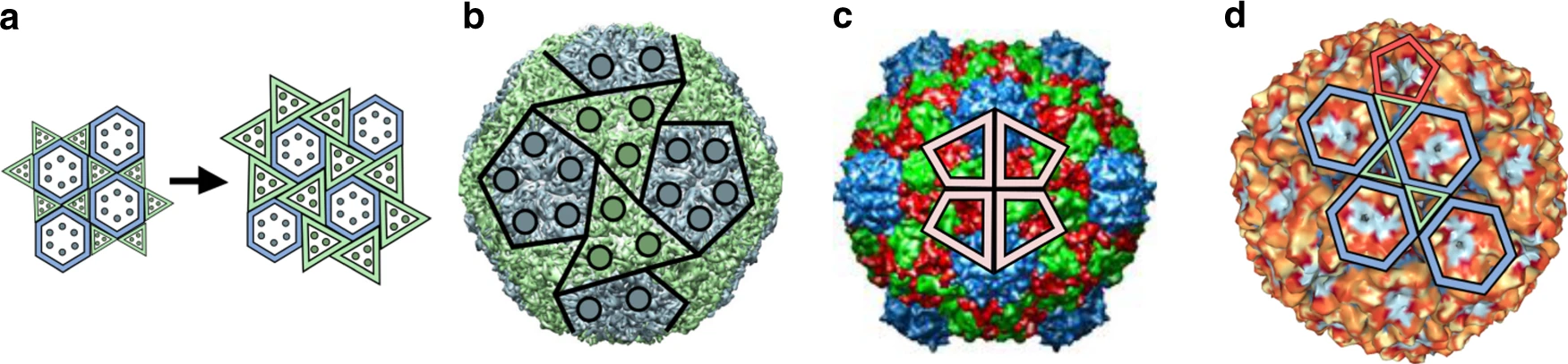
In larger viruses, the capsid shell often consists of more than one type of protein. Major capsid proteins can form the familiar icosahedral lattice while minor capsid proteins occupy a second set of configurations not captured by the original Caspar-Klug theory. Herpes simplex virus, for example, has a T=16 capsid, in which the major capsid protein forms pentamers and hexamers, while two other capsid protein types form groups of three molecules that sit in quasi-equivalent locations between them.
A Bacillus phage, with the ominous name Basilisk, is another well-studied case of this generalized quasi-equivalence. Even though such a complex capsid geometry comes at high additional genomic and assembly costs, its composition confers new properties, such as altered stability and different mechanisms of genome release within the host cells, potentially increasing their overall fitness.
Icosahedral Structures Beyond Viral Capsids
Even within this expanded repertoire of structures, icosahedral symmetry remains a constant, a remarkable testament to its apparent optimality as an evolutionary solution to efficient viral genome packaging and protection. Viruses across multiple, phylogenetically unrelated, families appear to have repeatedly rediscovered the icosahedral architecture, making it an exemplary case of convergent evolution that Conway Morris extolled and defended against the radical contingency view of Stephen Jay Gould.
The exact sequences of viral capsid proteins may vary widely, consistent with the “local” contingency of the evolutionary process. But even with this divergence, the tertiary and quaternary structures of capsid proteins and capsid structures of myriad viruses are remarkably similar, illustrating the “global” predictability of evolution. This predictability may be particularly defensible in the case of viruses, since they represent life stripped to the essentials of replication and assembly, and evolving under extreme geometric and genomic constraints.
But geometric constraints of this kind — when a sizable cargo needs to be efficiently packaged inside a container — seem to be at work not only in viruses but across a wide range of biological structures, from the molecular to organismal levels. Indeed, icosahedral symmetry has been found in bacterial microcompartments like carboxysomes (CO2 fixation organelles in cyanobacteria25), nanocompartments like encapsulins (self-assembling protein shells carrying molecular cargoes), lumazine synthase (a giant enzyme complex catalyzing riboflavin synthesis), and some of the unicellular eukaryotes with mineral skeletons like Radiolarians.26

Icosahedral architectures have also inspired countless artificial protein cages and self-assembling protein nanoparticles for drug delivery and vaccine design. Virus-like particles can self-assemble from proteins in cell culture and are completely harmless, since they lack the viral genome itself or carry a greatly modified version of it that eliminates virulent genes. They can be modified to express antigens on the surface of the capsid, mimicking the repetitiveness and geometry of natural host-pathogen interactions. This grants them high immunogenicity by effectively engaging the receptors of the antibody-producing B-cells in the host organism (which respond more strongly to repetitive antigens). Indeed, two recently WHO-approved highly efficient malaria vaccines, RTS,S/AS01 and R21/Matrix-M, are based on virus-like particles.
In drug delivery, both virus-like particles and artificial protein cages of similar design are used to carry therapeutic loads like enzymes, small molecules, or nucleic acids. Thanks to the relative ease of genetic engineering of viruses, it’s possible to generate libraries counting thousands of virus-like particles. These can be mutated or chemically modified on the surface, to ensure target specificity, or on the inside, to hold onto different molecular loads.
Fully de novo computationally designed protein cages can extend beyond what is typical in nature in terms of their stability, porousness, or even programmable disassembly in response to various conditions. Even so, the high symmetry and repetitive near-identical local interactions afforded by the icosahedral symmetry are still strongly favored in these novel engineered protein nanomaterials. Under exacting physical and informational constraints, both evolution and human engineering have converged on the same shape — a strong attractor in the space of biological containers.
{{signup}}
{{divider}}
Ulkar Aghayeva is a science writer and a columnist at Asimov Press. She also writes about science history on her blog Measure for Measure and about music history and cognition on The Bass Line.
Cite: Cite: Aghayeva, Ulkar. “Why Are Viral Capsids Icosahedral?” Asimov Press (2026). DOI: 10.62211/24ou-76ew
Footnotes
- This was illustrated vividly by Ray Bradbury in his wonderful short story A Sound of Thunder.
- As quoted in Keijzer FA (2017). Evolutionary convergence and biologically embodied cognition. Interface Focus 7: 20160123.
- Including both enveloped and non-enveloped capsids (with or without a cellular membrane around them, derived from the host cell). The rest are rod-like, helical (tobacco mosaic virus, rhabdoviruses and some other negative-strand RNA viruses), filamentous (Filoviridae like Ebola and Marburg virus), or have more complex capsid morphologies (e.g., ovoid virions of poxviruses).
- Though thanks to overlapping reading frames and alternative translation starts, those four genes produce seven proteins, including three surface antigens.
- In older papers on viral capsid structure, one can even find the term “spherical viruses.”
- Are there genome size limits for an icosahedral capsid geometry? One of the largest known viruses with a true icosahedral capsid is Megavirus chilensis with a genome of 1.26 MB and capsid size of ~440 nm. Mimiviruses also have genomes in the same range and icosahedral capsids of 200-400 nm surrounded by a fibrous layer. But even larger viruses, such as Pandoraviruses (2.5+ MB genome) and Pithoviruses (~1.5 μm capsid size) have alternative, more unusual morphologies to accommodate their large genomes.
- Nucleic acids are also bound by water molecules (hydrated), and the hydration forces, especially at high packing densities, add to self-repulsion and the resulting internal pressures exerted by the genome inside the capsid shell.
- In viruses with single-stranded RNA genomes, the capsid shells spontaneously assemble, simultaneously packaging their flexible and compact genome through binding of capsid proteins to specific sites. But dsDNA viruses assemble by first forming an empty capsid, and subsequently packaging their stiff and generally larger genome using strong ATP-driven molecular motors.
- Based on the work of Gregory J. Morgan, who wrote extensively on the history of the development of the Caspar-Klug theory.
- In 1954 he published a paper on the TMV structure with the help of Francis Crick, whose own interest in viruses developed during the war years. As it turned out, Watson erred in estimating the number of viral capsid proteins per helical turn and was later corrected by Rosalind Franklin.
- Also at Cambridge, Sydney Brenner and virologist Robert Horne developed a new method of “negative staining” that allowed for more fine-grained electron microscopic imaging of viral capsids, indispensable for later theory development on their structure. In negative staining, it is the imaging background that is stained, leaving the specimens untouched and visible by contrast (while in positive staining, the specimen itself is stained). In electron microscopy, negative staining involves applying a heavy metal salt like uranium acetate or phosphotungstic acid on the imaging grid, so that it strongly scatters electrons and appears dark. The specimen remains relatively electron-transparent and appears bright on the EM images. This method is especially well-suited for naturally low-density specimens like viral capsids.
- Caspar’s discovery was in part serendipitous, due to an unconventional positioning of his sample, which allowed him to observe a new and unexpected diffraction pattern. Five-fold symmetries are unexpected in a crystal structure since they cannot tile the 3D space. But this five-fold symmetry was not a feature of the crystal lattice consisting of viral capsids but rather a feature of the capsids themselves.
- It was in light of these experiments that Crick and Watson published their paper on the principle of genetic economy, discussed earlier. It’s worth mentioning that the paper was published before the genetic code was firmly established (implying the 3:1 ratio between nucleotides and corresponding amino acids), so Watson and Crick were taking some risk advancing this paper.
- Caspar took the responsibility of giving a conference talk that Franklin was scheduled to deliver later that year, and he initiated a co-authorship of the accompanying conference paper with Klug on X-ray diffraction studies of viruses. Caspar’s paper was a tribute to her work on the structure of TMV.
- See footnote 10.
- Despite being expelled from that university twice during his college years.
- More formally, if an icosahedron is flattened, it can be represented using a hexagonal tiling (honeycomb lattice), with each hexagon divided into six triangles. Euler’s theorem shows that in order to create a closed polyhedron, 12 of the hexagons from the lattice must be replaced by pentagons to make possible the 2D to 3D transition.
- Assuming quasi-equivalent connections, Caspar worked out a simple formula predicting the exact number of capsid subunits as T = h2 + hk + k2, where h and k are non-negative integers.
- More formally, one could apply a hexagonal lattice to the surface of an icosahedron, and replace each hexagon with six triangles.
- This remarkable prediction held for several decades until an exception to it was discovered in Caspar’s own lab. Some very large viral capsids still adhere to the capsid structure predicted by the Caspar-Klug theory, like the Paramecium Bursaria Chlorella Virus, with a T-number of 169.
- Another major source of inspiration for Caspar and Klug was Kenneth Snelson’s tensegrity sculptures — their mechanical stability is based on the same principles as the structural stability of viral capsids. Tensegrity is the integrity of structures under tension (flexible wires) and compression (rigid struts). Such structures are resilient to moderate deformation and maintain an equilibrium state in the absence of it. In an icosahedrally symmetrical tensegrity structure, the struts naturally arrange themselves into quasi-equivalent positions, as in viral capsids — again, demonstrating the same principles of organization spanning orders of magnitude of scale.
- Later, in 1985, another analogous structure was discovered by a group of chemists, including Harold Kroto, Robert Curl, and Richard Smalley at Rice University at the level of resolution even smaller than viruses. The 60-carbon cage-like molecule is known as buckminsterfullerene, or “buckyball.” With facets made up of hexagons and pentagons of carbon atoms, it looks like a soccer ball (a truncated icosahedron), recapitulating the geodesic dome architecture at the molecular level. (The three scientists shared the Nobel Prize in Chemistry in 1996 for the discovery of fullerenes.)
- Later it was shown that spontaneous assembly happens in single-stranded RNA viruses whereas more complex dsDNA viruses use molecular motors for loading the nucleic acids into the largely preformed protein capsid. See also footnote 7.
- Among other methods of making a Penrose tiling.
- Carboxysome is especially illuminating as it offers another instance of convergent evolution; the two lineages of carboxysomes (α and β) in marine and freshwater cyanobacteria evolved independently but share the same general structure, despite differences in gene organization and protein sequences. Only 100-500 nm in diameter, these icosahedral structures are composed of hexameric proteins, with pentamers at the vertices. They serve as reaction chambers for ribulose bisphosphate carboxylase/oxygenase (RuBisCO), the famous CO2-fixing enzyme, known for its catalytic slowness and poor selectivity for CO2 over O2. Inside carboxysomes, RuBisCO is colocalized with another enzyme, carbonic anhydrase, which converts bicarbonate into carbon dioxide. Thanks to the local abundance of CO2, RuBisCO is much more efficient than on its own. Carboxysome shells are also selectively permeable for oxygen, mostly keeping it out and preventing it from competing with CO2 for RuBisCO’s catalytic activity.
- One of them, helpfully named Circogonia icosahedra, indeed looks like a straightforward icosahedron, with some decorations affixed to its vertices. Though, to be fair, there are also straightforward octahedra (Circopurus octahedrus)and dodecahedra (Circorrhegma dodecahedra) among Radiolarians.
Always free. No ads. Richly storied.
Always free. No ads. Richly storied.
Always free. No ads. Richly storied.